Treating claudication and rest pain due to chronic total occlusion of the SFA
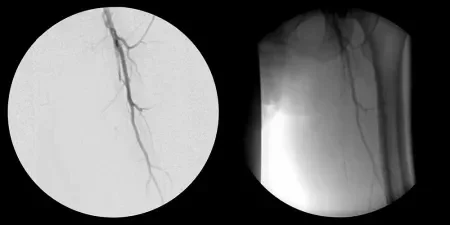
Challenge
- 65-year-old woman presented with severe peripheral arterial disease, claudication and rest pain of left lower extremity
- Angiography revealed total occlusion of the left superficial femoral artery (SFA)
Images courtesy of James Otto, M.D. Used with permission.
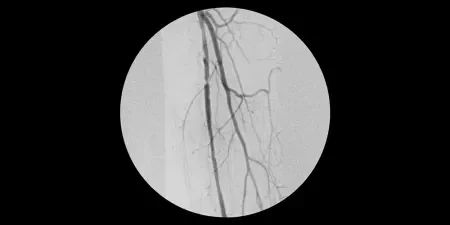
Procedure
- Obtained percutaneous access into the right femoral artery with ultrasound guidance. Then performed an angiogram of the left lower extremity.
- Crossed SFA chronic total occlusion (CTO) with 035 TERUMO® RADIFOCUS® GLIDEWIRE® ADVANTAGE and 035 TERUMO® NAVICROSS® Support Catheter
- Atherectomy completed with CARDIOVASCULAR SYSTEMS DIAMONDBACK 360® Peripheral Orbital Atherectomy System 2.0 Solid Crown
- Followed by angioplasty with a 5 mm percutaneous transluminal angioplasty (PTA) balloon
- Deployed two 6 mm x 15 cm GORE® VIABAHN® Devices in the SFA and post-dilated with 6 mm PTA balloon
Image courtesy of James Otto, M.D. Used with permission.
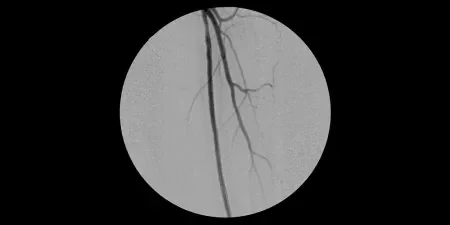
Result
- Completion angiogram showed excellent flow of the left lower extremity. Immediately post-op, complete resolution of left sided severe claudication.
- At follow-up office visit 2.5 weeks post-op, the patient presented with complete resolution of claudication and rest pain. Patient had palpable tibial pulses bilaterally
Image courtesy of James Otto, M.D. Used with permission.
Case takeaways
-
As demonstrated in this case, the GORE® VIABAHN® Endoprosthesis offers excellent patency in the treatment of long SFA total occlusions and should be considered first-line treatment for complex SFA disease

- GORE® VIABAHN® Endoprosthesis with Heparin Bioactive Surface [Instructions for Use]. Flagstaff, AZ: W. L. Gore & Associates, Inc; 2014. MD147177.
This content is for informational purposes only, is not advice or a guarantee of outcome. It is not a substitute for professional medical advice, diagnosis or treatment. Individual results and/or treatment may vary based upon the circumstances, the patient’s specific situation, and the healthcare provider’s medical judgment.