Device Specifications GORE® CARDIOFORM ASD Occluder

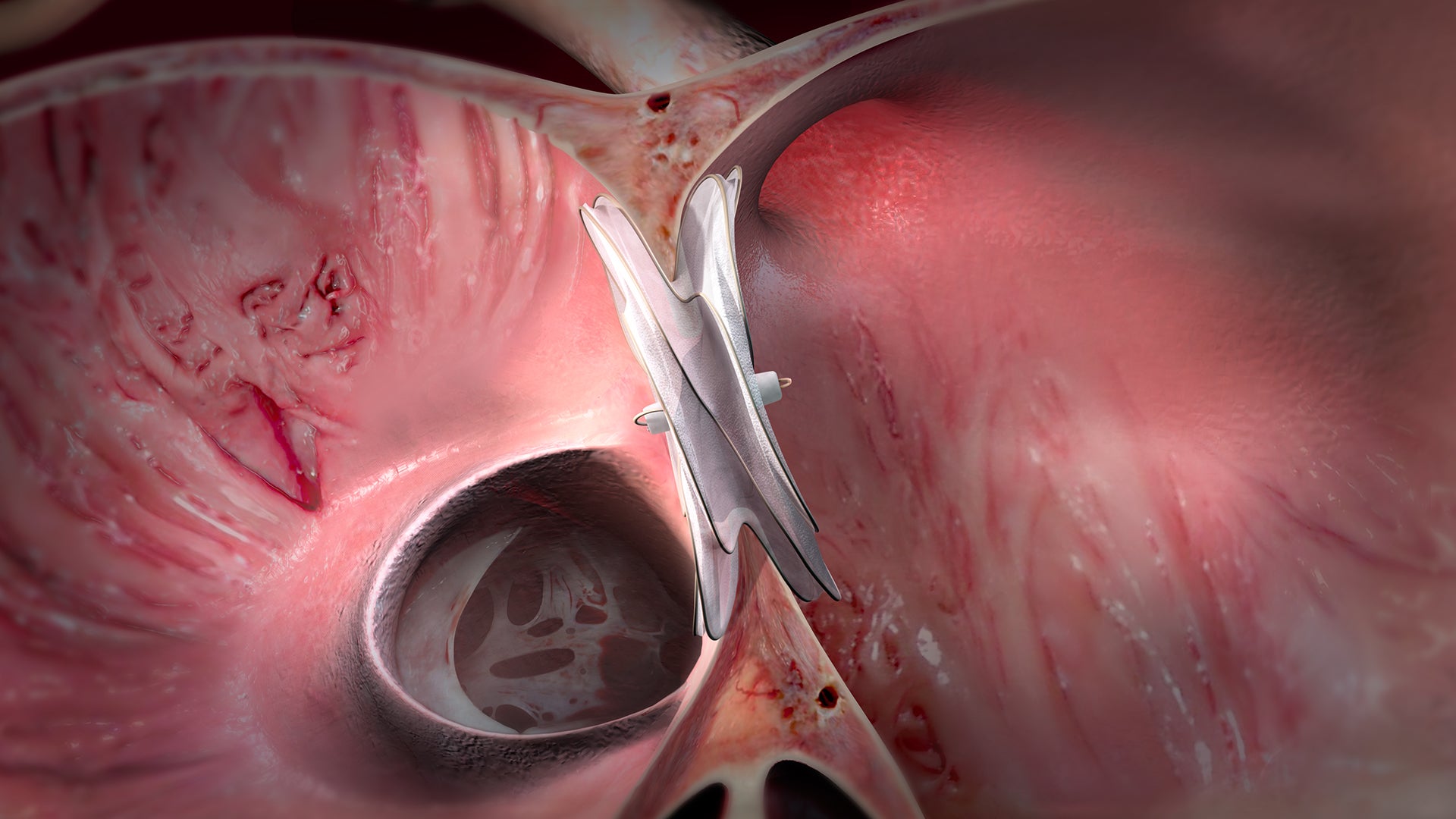
With the conformable design of the GORE® CARDIOFORM ASD Occluder, five catalogue numbers cover ASDs from 8 to 35 mm.*

| Device size (Disc diameter) | Treatment range measured with stop flow balloon sizing | Sheath size† | U.S. catalogue number‡ |
|---|---|---|---|
| 27 mm | 8-15 mm | 10 Fr | ASD27A |
| 32 mm | 13-20 mm | 10 Fr | ASD32A |
| 37 mm | 18-25 mm | 11 Fr | ASD37A |
| 44 mm | 23-30 mm | 12 Fr | ASD44A |
| 48 mm | 28-35 mm | 14 Fr | ASD48A |
* Refer to Instructions for Use at eifu.goremedical.com for a complete description of all applicable indications, warnings, precautions and contraindications for the markets where this product is available. RXOnly
† Recommendation for sheath size is 2 Fr larger when used with a wire.
‡ Catalogue numbers may vary by country or region.

Refer to Instructions for Use at eifu.goremedical.com for a complete description of all applicable indications, warnings, precautions and contraindications for the markets where this product is available. RXOnly
INDICATIONS FOR USE IN THE U.S.: The GORE® CARDIOFORM ASD Occluder is a permanently implanted device indicated for the percutaneous, transcatheter closure of ostium secundum atrial septal defects (ASDs).
CONTRAINDICATIONS: The GORE® CARDIOFORM ASD Occluder is contraindicated for use in patients: Unable to take anti-platelet or anticoagulant medications such as aspirin, heparin or warfarin; with anatomy where the GORE® CARDIOFORM ASD Occluder size or position would interfere with other intracardiac or intravascular structures, such as cardiac valves or pulmonary veins; with active endocarditis, or other infections producing bacteremia, or patients with known sepsis within one month of planned implantation, or any other infection that cannot be treated successfully prior to device placement; with known intracardiac thrombi.